|
Volume VII No. 2 - August, 1934
The Geology Of The Garfield Trail
By Carl R. Swartzlow, Ranger-Naturalist

The Garfield Trail speaks in many languages. The song of the birds
and the rustle of the breezes through the hemlocks and pines are the
first sounds to great the ear, and they follow one all the way. This
language is also expressed in less musical tones. As one approaches the
Rim and looks toward the Lake, a rumbling sound is heard and attracts
the attention to boulders, loosened by erosion, tumbling to the narrow
beach at the water's edge. Jets of dust rise at points where the
boulders strike on their downward journey. It is only after gazing at
the inner Rim for several minutes that the grandeur of the scene unfolds
and one realizes that the landscape of color dominates this trailside.
The Garfield Peak Trail may be called a study in brown. From the
point where one first ascends the trail, thence to the top of the peak,
one is constantly impressed by the ever changing shades of red, brown,
and yellow. The causes for this particular series of colors are related
to the most fundamental processes of geology an should be a part of the
knowledge of every lover of the out of doors.
The reds and browns of the rocks along the trail are usually a
result of the first stages of rock decay, and all subsequent compounds
released are stained with these colors. The constituents of the
pigments are oxygen, iron, and water. The iron oxides (iron united with
oxygen) are a constituent of nearly every variety of igneous rock. Iron
may not be a diagnostic element in some varieties of rock but its
presence is all but universal. The abundance of iron oxide determines
in a large measure the depth of color found in the weathered rock
products.
Usually the iron in fresh rocks is not fully united with oxygen,
i.e., oxidation is not complete and in this state the iron is soluble
and is relatively colorless. Moisture that comes in contact with rocks
has previously dissolved varying amounts of the atmospheric gases, of
which oxygen is a common constituent. The oxygen in the water unites
with the iron of the rocks and produces a compound in which the iron has
taken on all the oxygen possibly and in this latter state is one of
nature's most insoluble compounds. If water as such has united with the
iron oxide the resulting compound (the mineral limonite) has a brown
color, and if the compound is diluted the resulting color is yellow. If
no water has entered into the reaction, the color of the resulting rock
is red - the mineral hematite. If the soluble and insoluble iron
compounds are mixed a greenish color is produced. It can be seen
readily that combinations of these colors in various stages of dilution
can produce an infinite variety of shades that enhance the beauty of the
trailside.
At the beginning of the Garfield Trail one walks over a mass of
buff-to-tan pumice dust. Undecomposed fragments of this volcanic glass
reflect the sunlight as if the pumice contained myriads of diamond
chips. In time their lustre will be dulled by the chemical action of
the atmosphere or by organic acids released by decaying vegetation. The
pumice soils contribute little essential plant food, and only the more
hardy grasses and flowers are found growing upon them. Where abundant
vegetation appears to be growing in pumice, the plants are usually
rooted in more fertile soils below.
A few yards beyond the point where the trail first touches the Rim,
and at several other points along the trail, the lava rocks (mainly
andesite agglomerate) have been decomposed and young soils have been
formed. These soils have a chalky appearance in contrast to the usual
yellow or brown soil along the trail. These formations are seldom a
result of normal weathering processes, but are probably due to the
action of heated waters that escaped along the slopes of Mt. Mazama.
The rocks have been almost completely decomposed. The residual material
is the mineral kaolin or some variety of it. (Kaolin is the chief
mineral constituent of clay). If one moistens his fingers and rubs them
over some of these particles, a greasy or doughy ball of clay is readily
formed.
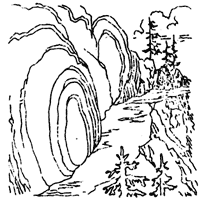
About halfway to the top of the Peak there are additional examples
of rock weathering that are more common but are none the less
interesting. Large masses of volcanic agglomerate have been weathered
for long periods of time by normal processes. In the construction of
the trail, cuts have been made through the rock mass, and cross sections
of many boulders are left to tell the steps that nature has used to
bring about the changes from solid rock to soil. The sub-angular
fragments of rock have clear outlines, but they can be crushed easily
with the fingers.
In many cases a series of concentric bands, similar to the peelings
of an onion, surround a central portion of rock that is relatively
undecomposed. The cause of the banding is a common process observed in
moist climates. Moisture penetrates the pore spaces of the rock. The
depth of penetration depends upon the size of the pores, temperature,
and character of the solutions. When the optimum depth has been
reached, the moisture tends to decompose the rock. The new products
formed are of greater volume than the original rock minerals.
Consequently, swelling occurs and the shell of altered rock, as thick as
the depth of moisture penetration, cracks away from the original rock.
After the first shell has been released there is a ready passage for the
succeeding influxes of moisture to penetrate the rock below and the same
process repeats itself. This type of rock decay is called spheroidal
weathering. Upon prolonged exposure to the elements the banding
disappears and a homogeneous mass of soil is found where the boulder was
situated originally. In a few instances the bands may appear to be of
different colors. This is probably due to the amount of iron oxide
absorbed by the soil during the breakdown of the boulders.
The foregoing examples can be readily contrasted with the fresh
unaltered rocks along the talus slopes and the rock cuts along the
trail. One is impressed by the ceaseless effort of natural forces to
break down the rocks on the earth's surface. This is to provide soil
and plant food so that the fauna and flora of the earth may carry on
their life functions.
The names of several rocks have been mentioned above. The first,
pumice, is present in varying amounts along most of the trail. Its most
common mode of occurrence here is as a fine buff-colored material. In a
few places fragments several cubic inches sin volume may be found.
These larger fragments exhibit all of the common characteristics of
pumice; namely, light buff-to-tan color, glassy texture, and high
porosity.
Pumice is invariably associated with explosive vulcanism and
consequently the original magma is charged with gases, usually water
vapor and carbon dioxide. The gases, under pressure, are forced into
the viscous lava, thus producing the characteristic porous texture. The
lava hardens before the pore spaces are eliminated.v
The next most abundant rock is andesite. It differs markedly from
pumice, both in composition and the manner of its formation. Andesite,
the common flow rock of Crater Lake, is rich in iron, magnesium, and
calcium, and poor in silica. Pumice, on the other hand, has a high
silica content with very minor amounts of the other elements. The
pressure of the iron, magnesium, and calcium in the lavas increases
their fluidity, thus permitting them to flow over wide areas. Common
with most andesites, those of Crater Lake are porphyries. That is,
there are large crystals embedded in a more dense background called the
ground mass. The most common cause for the formation of porphyries is a
change in the rate of cooling. When deep within the earth, the lava
started to solidify or crystallize and numerous minerals (feldspars)
grew to the size shown in the rocks. Then pressure from below forced
the lava to the cool surface where rapid solidification stopped the
mineral growth and caused the running lava to harden about the earlier
formed minerals.
The only other rocks of importance is a volcanic agglomerate. This
is a rock composed of fragments of the various igneous (lava) rocks of
the region. During periods of explosive action all of the types of rock
present were thrown into the air and then upon descent filled cracks and
gullies in the sides of Mt. Mazama. Later, lava flows or percolating
waters caused the fragments to be more or less consolidated. Two-thirds
of the way up the trail an excellent view station is located where one
can observe masses of boulders caught in the lavas. This shows that in
some parts of Mt. Mazama, lava flows and explosive eruptions were
simultaneous. Perhaps the boulders merely tumbled down the mountain
side and were caught in the flow, or else were engulfed as the lava
moved along.
Fragments of the mineral quartz may be seen among the talus debris
along the trail. The mineral is a variety known as milky quartz and is
common the world over. It has formed by solutions rich in silica rising
through cracks in the sides of Mt. Mazama. The rarity of quartz as well
as other secondary minerals is significant.
In several places along the Garfield Peak Trail are large white
blotches on the rocks. This is especially true of the areas of volcanic
agglomerate. These white spots represent areas where hot moist gases
escaped to the surface of Mt. Mazama.
The rocks are largely kaolinized, but with the outline of the rock
fragments retained. If the rocks are rubbed with the fingers the
typical clayey feel can be readily recognized.

| 
