|
NATIONAL PARK SERVICE
Mountain Goats in Olympic National Park: Biology and Management of an Introduced Species |

|
| Mountain Goat Herbivory |
CHAPTER 12:
Rare Plants
E. G. Schreiner, M. B. Gracz, T. N. Kaye, A. Woodward, and N. M. Buckingham
Rare plants—about 9% of the —1,200 native vascular flora—contribute measurably to the biodiversity of the Olympic Peninsula, The Washington State Natural Heritage Program lists 395 vascular plant species and varieties as rare in Washington State (Washington Natural Heritage Program 1990)—106 (27%) of these occur on the Olympic Peninsula, an area representing less than 8% (13,800 km2) of the surface area of the state. Olympic National Park has an even higher concentration of these plants with 77 species (19%) on just over 2% (3,700 km2) of the state.
Concern for the rare flora of Olympic National Park resulted from the discovery that some endemic plants15 were consumed by mountain goats. Pike (1981) found that three endemics exhibited increased standing crop when they were protected from ungulates in goat summer range. Fragments of the endemic Campanula piperi were also detected in goat fecal samples (see Chapter 6). The detection of this species in goat diets was remarkable because of the plant's limited occurrence and low standing crop. Additionally, direct observations and permanent plots indicated that goats consumed and sometimes wallowed on the endemic Astragalus australis var. olympicus.
15See Chapter 3 for a discussion of endemism and a complete list of endemic taxa.
To assess the risk posed by mountain goats to the long-term persistence of rare plants, we (1) made a thorough search of several herbaria for records of plant locations, (2) compiled information from historical and modern plant lists, (3) examined all sample plot data for the presence and location of the Washington Natural Heritage Program plants, (4) conducted a survey for rare plants at high elevations, and (5) studied the habitat requirements and demography of Astragalus australis var. olympicus.
Distribution of Rare Plants
Rare plants listed by the Washington Natural Heritage Program occurred throughout the Olympic Peninsula (Fig. 39). Most taxa were evidently rare locally—30 were known from only 1 area and 12 from only 2 areas on the peninsula. Particularly high concentrations of rare plants occurred near sea level at Lake Ozette (14) and at high elevations in the northern and northeastern part of the peninsula: Mount Angeles (22), Elk Mountain (14), Blue Mountain (17), Royal Basin (16), and in the Buckhorn Mountain region (26) on Olympic National Forest.
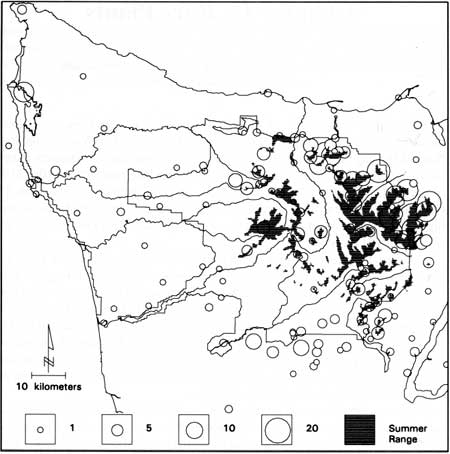
|
| Fig. 39. Locations of rare vascular plants listed by the Washington Natural Heritage Program (1990) in relation to mountain goat summer range. Circles are proportional to the number of taxa in each area. |
The elevation distribution of rare plants was bimodal (Fig. 40A). Thirty-eight taxa had average elevations below 305 m, 27 between 305 and 1,520 m, and 37 greater than 1,520 m. Forty-three percent of the 77 rare plants found in the park occurred within mountain goat summer range (i.e., average elevation above 1,520 in; Table 33). Nine high-elevation taxa were known from only one park location, and 17 occurred at 5 or fewer sites in the park. Seven of the eight Olympic Peninsula endemics had average elevations between 1,500 and 1,870 m, and one, Erigeron peregrinus ssp. peregrinus var. thompsonii, occurred at low elevations (Fig. 40B). Additionally, three of the Olympic Peninsula-Vancouver Island endemic taxa had average elevations higher than 1,520 m.
Table 33. Olympic Peninsula vascular plants listed by the Washington Natural Heritage Program (WNHP; 1990) that have average elevations within mountain goat (Oreamnos americanus) summer range in Olympic National Park (ONP).
| Species (WNHP status)a |
Totalb | Average elevation (m) | Pc | Bc | Oc | Uc |
| Species known from 5 or fewer ONP locations | ||||||
| Botrychium lunaria (3) | 1 | 1,768 | 1 | 0 | 0 | 0 |
| Douglasia nivalis var. dentata (4) | 1 | 1,829 | 1 | 0 | 0 | 0 |
| Salix brachycarpa ssp. b. var. b. (4) | 1 | 1,905 | 1 | 0 | 0 | 0 |
| Dryas drummondii var. d. (3) | 1 | 2,057 | 1 | 0 | 0 | 0 |
| Pellaea breweri (3) | 2 | 1,631 | 1 | 0 | 1 | 0 |
| Poa rupicola (4) | 2 | 1,846 | 1 | 0 | 1 | 0 |
| Botrychium pinnatum (3) | 3 | 1,591 | 1 | 0 | 2 | 0 |
| Carex obtusata (3) | 5 | 1,800 | 1 | 0 | 4 | 0 |
| Zigadenus elegans ssp. e. (4a) | 6 | 1,795 | 1 | 0 | 5 | 0 |
| Poa suksdorfii (4) | 3 | 1,788 | 2 | 0 | 1 | 0 |
| Saxifraga debilis (3) | 3 | 1,798 | 2 | 0 | 1 | 0 |
| Erigeron flettii (4)d | 19 | 1,580 | 2 | 3 | 14 | 0 |
| Phlox hendersonii (4) | 10 | 1,908 | 4 | 0 | 6 | 0 |
| Lloydia serotina ssp. s. (4) | 11 | 1,676 | 4 | 0 | 7 | 0 |
| Astragalus australis var. olympicus | 5 | 1,554 | 4 | 0 | 1 | 0 |
| Asplenium viride (4) | 8 | 1,696 | 5 | 1 | 2 | 0 |
| Saxifraga tischii (4a)e | 8 | 1,856 | 5 | 1 | 2 | 0 |
| Species known from 6 or more ONP locations | ||||||
| Oxytropis viscida var. v. (3) | 6 | 1,734 | 6 | 0 | 0 | 0 |
| Poa arctica ssp. grayana (3) | 8 | 1,539 | 6 | 0 | 2 | 0 |
| Arnica nevadensis (4) | 10 | 1,759 | 8 | 1 | 1 | 0 |
| Salix arctica (4) | 10 | 1,789 | 8 | 0 | 2 | 0 |
| Poa paucispicula (4) | 10 | 1,687 | 9 | 0 | 1 | 0 |
| Carex raynoldsii (4) | 11 | 1,667 | 6 | 0 | 5 | 0 |
| Synthyris pinnatifida var. lanuginosa (3)d | 14 | 1,867 | 8 | 1 | 5 | 0 |
| Arnica rydbergii (4) | 16 | 1,676 | 10 | 0 | 6 | 0 |
| Geum triflorum var. campanulatum (4) | 18 | 1,654 | 11 | 0 | 7 | 0 |
| Erigeron compositus var. discoideus (4) | 18 | 1,784 | 13 | 0 | 0 | 5 |
| Senecio neowebsteri (4)d | 23 | 1,824 | 16 | 1 | 6 | 0 |
| Orthocarpus imbricatus (4) | 24 | 1,683 | 16 | 1 | 6 | 1 |
| Viola flettii (4)d | 36 | 1,751 | 19 | 3 | 13 | 1 |
| Petrophytum hendersonii (4)d,f | 46 | 1,500 | 24 | 2 | 17 | 3 |
| Campanula piperi (4)d | 47 | 1,698 | 27 | 4 | 16 | 0 |
| Aster paucicapitatus (4)e | 47 | 1,574 | 34 | 1 | 11 | 1 |
| Pedicularis bracteosa var. atrosanguinea (4)e,g | 4 | 1,547 | 4 | 0 | 0 | 0 |
| Species that may occur in ONP | ||||||
| Senecio lugens (4) | 8 | 1,769 | 0 | 1 | 7 | 0 |
| Drabaton gipes (3) | 1 | 1,890 | 0 | 1 | 0 | 0 |
| Species not known to occur in ONP | ||||||
| Draba cana (3) | 1 | 1,829 | 0 | 0 | 1 | 0 |
| Astragalus microcystis (3) | 2 | 1,813 | 0 | 0 | 2 | 0 |
aWashington Natural Heritage Program categories: (2) threatened; (3) sensitive; (4) monitor; and (4a) monitor for listing. bTotal = number of areas where the plant is known to occur on the Olympic Peninsula. cP = number of known Olympic National Park locations; B = number of known locations on the border with the park (i.e., may or may not be in the park); 0 = number of known locations outside the park; U = precise location unknown (e.g.. Clallam County). dOlympic Peninsula endemic. eOlympic Peninsula—Vancouver Island endemic. fWe listed this species as occurring in goat summer range because it occurs mainly on rocky outcrops. gIncomplete data. | ||||||
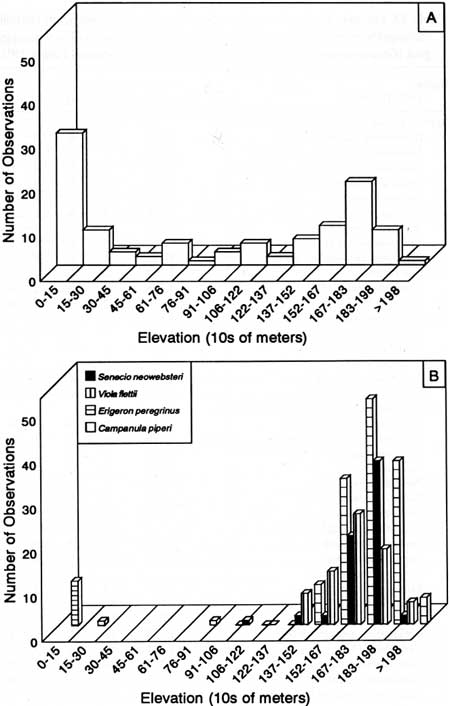
|
| Fig. 40. Elevation distribution of rare plants on the Olympic Peninsula. A taxa listed by the Washington Natural Heritage Program (1990); B = selected taxa endemic to the peninsula. |
Rare Plants and Mountain Goats
Goat censuses showed that goat summer range overlapped rare plant distributions (Fig. 39). Evidence of mountain goat use was still visible in 1988 and 1990 even though the goat population had decreased from an estimated 1,175 ± 171 (SE) in 1983 to 389 ± 106 animals in 1990. Twenty-one of 22 areas surveyed contained rare plants, and all areas exhibited evidence of mountain goats (see Chapter 9). However, the extent of herbivory by mountain goats on rare plants was difficult to assess because so few rare taxa occurred in plots.
Grazing on two endemic taxa, Aster paucicapitatus and Astragalus australis var. olympicus, was attributed to mountain goats (Table 34). Aster paucicapitatus was grazed in 12 of the 18 plots where it occurred in Royal Basin, a medium goat density area, but was not grazed in the 21 low-density-area plots at Grand Pass. Because deer and marmot signs were encountered in both areas and goat sign was far more prevalent at Royal Basin than Grand Pass (see Table 24), goats were implicated as responsible for the grazing (elk sign was not observed in either area). Aster paucicapitatus was one of 33 species considered to be forage selected by mountain goats (see Table 27). Numerous goat wallows, tracks, and pellet groups in plots containing Astragalus australis var. olympicus indicated that mountain goats grazed this species. Astragalus was grazed in all three medium-goat-density areas where it occurred.
Table 34. Average grazing, plant and grazing frequency, and species
relative grazing frequency (SRGF) of rare vascular plant taxa listed
by the Washington Natural Heritage Program (1990) in subalpine plots
of Olympic National Park.
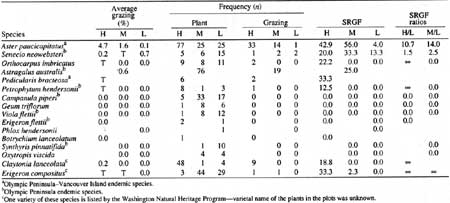
(click on image for a PDF version)
Fourteen other rare taxa were found in plots used to describe subalpine and alpine plant communities (Table 34) but were not sufficiently abundant to make an evaluation of goat effects on them. Campanula piperi, for example, occurred in only 5 plots in high, 33 plots in medium, and 17 plots in low goat density areas. The plants were not grazed in any area, which may indicate that Campanula was nonselected by goats or that we did not have enough plots in high density areas to detect grazing.
Similarly, permanent plots in the three areas where goat densities were reduced (see Chapter 10) contained four rare plant taxa. However, these too were so infrequent that the effects of goat herbivory could not be established (Schreiner et al. 1993).
Astragalus australis var. olympicus: A Case Study
Astragalus australis var. olympicus is a rare endemic known only from the dry northeastern Olympic Mountains. The metapopulation of 3,800-4,000 individuals occurs at 1,525-1,825 m in Olympic National Park as 11 subpopulations (Kaye 1989).16 Habitat includes south-facing subalpine talus, windswept ridges, and knolls on calcium-bearing substrate. This taxon is listed as threatened by the Washington State Natural Heritage Program (1990) as well as a Category 1 candidate for the federal endangered species list (U.S. Fish and Wildlife Service 1993). Observations of mountain goat grazing, trampling, and wallowing in A. australis var. olympicus subpopulations prompted concern for the well-being of this plant.
16A new subpopulation outside the park was recently (July 1992) discovered in the Mount Townsend area on Olympic National Forest.
Astragalus australis var. olympicus or its precursor may have reached the Olympic Mountains from the north. The distribution of A. australis is similar to that of another Olympic Mountain rare plant, Oxytropis viscida var. viscida. Barneby (1964) suspected they followed the same migration routes and that both taxa reached the Olympic Peninsula via Beringia and then south during a Pleistocene interglacial period. Astragalus australis var. olympicus probably did not arrive here by long-distance dispersal; the closest population of A. australis var. glabriusculus (the nearest relative of A. australis var. olympicus) is about 500 km distant in northeastern Oregon.
Isolation in the Olympic Mountains left a population of A. australis with unique traits. The inflated seed pod of var. olympicus sets it apart from var. glabriusculus, though the pods of some Alaskan forms also are "decidedly tumid" (Barneby 1964). Although var. olympicus has not been isolated long enough to evolve into a separate species, as have some other Olympic endemics, the distinct inflated pods could represent an important dispersal adaptation. Seeds from flat pods can be expelled explosively, be eaten, or simply drop from the plant, but inflated legume pods can also be dispersed by wind (Ridley 1930). With further isolation, this variety may evolve into a separate species, as speciation through reproductive isolation is fairly common in montane floras (Hickman 1968; Favarger 1972; Ives 1974; Stebbins 1982). Astragalus australis var. olympicus and associated species fit rather loosely into the cushion plant community described by Kuramoto and Bliss (1970). Species common to cushion plant communities and often found growing with Astragalus included Phlox diffusa, Festuca idahoensis, Allium crenulatum, and Lomatium martindalei (Kaye 1989). Kaye (1989) identified seven plant community types with Astragalus as a constituent. A feature held in common at all sites was calcareous substrate with pH almost always over 6.
Population Dynamics and Goat Effects
Intensive sampling of the physical and biological environment of Astragalus subpopulations was conducted at Blue Mountain and Mount Angeles where most of the plants occurred (Appendix A1). Plots were located in areas of physical site homogeneity that included at least one Astragalus individual and were distributed over the range of environmental conditions observed in each plant subpopulation. Ten plots were permanently marked, all Astragalus plants were mapped and counted, and grazing or physical injuries were noted from 1985 to 1991. Plots were distributed as follows: Unicorn Peak (2), Griff Peak (1), Blue Mountain (5), and Hurricane Hill (2). All mountain goats were eliminated from Blue Mountain, and goat density was reduced by 98% at Mount Angeles during 1981-89.
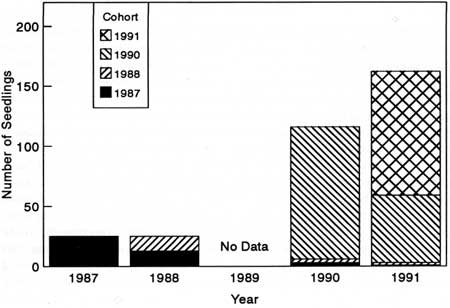
We counted 1,442 Astragalus individuals (36% of the estimated population) in 79 plots. Permanent plots revealed that the Astragalus subpopulations were more or less stable from 1985 to 1991 (Fig. 41) and that seedling establishment was highest in 1990 and 1991 (Fig. 42). Seedling mortality was about 50% per year in 1987-88 and 1990-91.
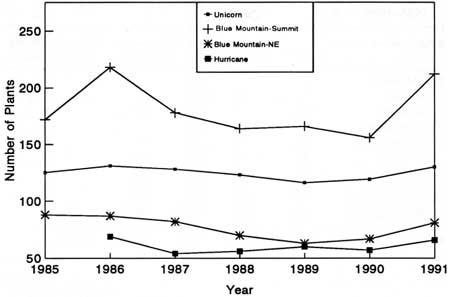
|
| Fig. 41. Total nonseeding Astragalus individuals in plots within four subpopulations. |
|
| Fig. 42. Total Astragalus seeding establishment and cohort survival in plots within four subpopulations. |
Mountain goat sign was found inside and adjacent to plots established in Astragalus subpopulations (Table 35). Astragalus was grazed in more than half of the subpopulations. Goat activity was particularly evident in the Mount Angeles Astragalus subpopulations, where sign was found inside or near 70% of the plots even though the goat population there had been substantially reduced.
Table 35. Evidence of mountain goat (Oreamnos americanus) use in and around plots established in subpopulations of Astragalus australis var. olympicus, 1985-1986.
| Subpopulation name |
Plots | Wallows within 50 m of plot |
Pellet groups inside plots |
Plots with grazing on Astragalus |
| Blue Mountain | ||||
| Summit | 6 | 10 | 9 | 0 |
| Northeast | 21 | 7 | 0 | 3 |
| Mount Angeles | ||||
| Northwest | 17 | 27 | 0 | 8 |
| First Top | 6 | 0 | 3 | 0 |
| Heather Park | 6 | 1 | 0 | 3 |
| Second Top | 14 | 31 | 10 | 3 |
| Unicorn/Griff Peak | 3 | 11 | 0 | 2 |
| Hurricane Hill | 6 | 0 | 0 | 1 |
| Total | 79 | 87 | 22 | 20 |
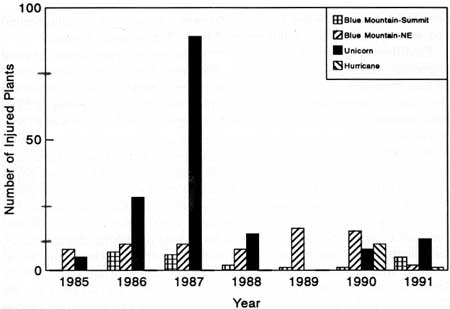
Permanent plots showed that Astragalus plants were injured (grazed or trampled) by goats. Injury was sporadic but sometimes intense (Fig. 43). Some injuries were obviously caused by goats (i.e., abundant goat sign adjacent to grazed or trampled Astragalus plants), but the cause of other injuries could not be determined. Goat wallowing in three plots was responsible for four dead plants in 1986. Extensive grazing and trampling of plants at Unicorn Peak in 1987 were entirely attributable to mountain goats. In the plots where most of the injuries took place, 26 of 50 subplots contained goat tracks, Grazing was evident on 72% (89) of the individuals in the plots—most were noted as 76-100% grazed. Goats evidently moved through the area and stopped to graze and wallow in the Astragalus.
|
| Fig. 43. Total number of grazed or trampled Astragalus in plots within four subpopulations during 1985-91. |
Astragalus Habitat
We performed three analyses to determine how much Astragalus habitat was present and how much was occupied in Olympic National Park. First, 687 of the plots used to describe subalpine and alpine vegetation17 (including 79 with Astragalus) were classified into communities based on plant cover (Appendix A1).
17Plots used in these analyses did not include those established in 1988 and 1990—hence the number of plots does not match those in Chapter 9.
Second, we employed nonparametric discriminant analysis to classify plots by similarities among environmental variables (e.g., slope, aspect, and rock fragment size). The plant community classification was then compared to the classification based on environmental variables. Agreement between these two classifications (i.e., plots were placed in similar groups by environmental- and plant-based methods) was assumed to mean that environmental variables adequately explained the plant community classification. We further assumed that if the plant-based classification grouped the plots that contained Astragalus. then a distinct set of environmental variables represented Astragalus habitat. Consequently, if the classification using environmental variables also grouped Astragalus plots, then Astragalus habitat was probably represented by that group of environmental variables. Finally, we used logistic regression to determine which environmental variables contributed most to explaining differences between plots containing Astragalus and those that did not.
The plant community classification resulted in four major groups that were readily discernible on the basis of plant composition—one group was composed entirely of Astragalus plots.18 The four groups reflected the importance of moisture and disturbance levels to subalpine community composition (Kuramoto and Bliss 1970; Pfitsch and Bliss 1985). All plots at the dry extreme (44) contained Astragalus, a slightly wetter group contained 44% (35) of the Astragalus plots, and 284 additional plots were dominated by species such as Phlox diffusa and Festuca idahoensis. Two groups were dominated by Carex spectabilis, Phyllodoce empetriformis, or Cassiope mertensiana.
18See Appendix A2 for a more detailed discussion of subalpine plant communities.
Astragalus habitat was limited in extent and may have been fully occupied. Discriminant analysis based on environmental variables classified Astragalus plots in groups that were comparable to the groups produced by the classification based on plant cover. The two methods classified 84% of the Astragalus plots similarly. Only 16% of the plots that did not contain Astragalus were classified by the discriminant analysis as having environmental conditions potentially suitable for Astragalus. Plots that lacked Astragalus but had Astragalus-like habitat occurred in wetter areas of the park (i.e., well outside the dry northeastern Olympic Mountains where the Astragalus occurs) and were apparently unsuitable or too distant for propagules to reach.
Environmental variables determined by the logistic regression to be most important in distinguishing Astragalus plots were slope, aspect, percent cover of bare mineral soil, total rock, and the cover of rocks in the >2-mm—4-cm size-class. Average slope of Astragalus plots was 28°, aspect averaged 201°, bare mineral soil averaged 11.9%, and average total rock cover was 81.9%. Average cover of rocks in the >2-mm—4-cm size-class was 56.3%.
Blue Mountain plots contained an average of 3 times more Astragalus plants per square meter than did plots representing Mount Angeles subpopulations (1.01 vs. 0.31; P < 0.01, Mann-Whitney U-test). The logistic regression equation describing Astragalus habitat characteristics was used to test the hypothesis that habitat differences were responsible for different densities of plants between the two areas. Based on habitat characteristics, the equation predicted that the mean probability of a plot containing Astragalus at Blue Mountain was 0.47 and 0.53 at Mount Angeles (P = 0.40, Mann-Whitney U-test). The major difference between the two areas was the historical density of mountain goats (much greater on Mount Angeles) rather than habitat characteristics.
Interpretation
These preliminary investigations of rare plants in the presence of mountain goat herbivory were inconclusive, Rare plants in the park are indeed rare—some occur in just one site, others occur as a few widely-spaced individuals. Our studies demonstrated that there is considerable overlap between mountain goat summer range and the habitat of rare plants. Even though we were unable to quantify the risk mountain goats pose to the persistence of rare plant populations, it is clear that mountain goats do affect and kill individual plants through grazing, wallowing, and trampling. Four endemic species were grazed by goats—one was considered a selected forage.
The Astragalus australis var. olympicus population trend shown by permanent plots represented only a short-term picture. Circumstantial evidence from habitat analysis suggested that goat herbivory may have reduced plant densities on Mount Angeles compared to Blue Mountain. Seedling establishment varied markedly among years. One or 2 years of successful seedling establishment means little to the persistence of a subpopulation or to species survival because of high mortality. Considerably longer study will be required to determine population trends. Goat effects on Astragalus were sporadic, sometimes intense, and had unknown long-term consequences to the survival of the species. We have insufficient data to cleanly reject the hypothesis that mountain goat herbivory does not jeopardize the persistence of rare plant populations. Enough information exists to be concerned about the long-term fate of these taxa. We do not expect goats to necessarily consume or trample the last individuals in a population of rare plants, but they may increase risk of extinction by fragmenting populations or habitats. Risk of extinction increases as populations are fragmented or reduced in number (Pimm 1991). Extinction often involves a combination of forces acting on small, fragmented populations (e.g., Interagency Spotted Owl Committee 1990). Assessment of the potential for long-term (i.e., 15-25 years or more) persistence of populations of individual taxa will require intensive study of plant demography and available habitat.
Finally, it is preposterous to expect all rare plants to respond similarly to goat herbivory. Definitions of rarity are based on abundance, habitat requirements, and distribution (Rabinowitz 1981) rather than autecological characteristics. Rare plants of the Olympic Mountains include annuals (Orthocarpus imbricatus), rosette-forming perennials occurring in meadows (Geum triflorum var. campanulatum), and perennials confined to rocky outcrops (Petrophytum hendersonii). Harper (1977), Grime (1979), and many others have expended considerable energy to demonstrate how autecological characteristics govern plant demography. This reinforces the need for long-term demographic studies of individual taxa—truly a formidable task considering the number of rare plants in the Olympic Mountains.
| <<< Previous | <<< Contents >>> | Next >>> |
chap12.htm
Last Updated: 12-Dec-2007